Products
- The on-target rate of pathogens are significantly influenced by the amount of pathogens within the samples. As shown in Figure, under the same host background, as the microbial reference material is gradually diluted, the amount of data decreases proportionally, along with a decrease in on-target rate.
-
Although multiple capture rounds can substantially improve on-target rate, this strategy comes with the risk of information loss (dropout). Especially when the amount of pathogen is low, the on-target rate is low and is more l sensitive to dropout. Therefore, careful consideration of the pros and cons is needed when contemplating multiple capture rounds. Additionally, from another perspective, when the pathogen genome copy numbers in the sample are in the single digits, increasing on-target rate merely involves repetitive sequencing of these few copies, yielding limited additional useful information.
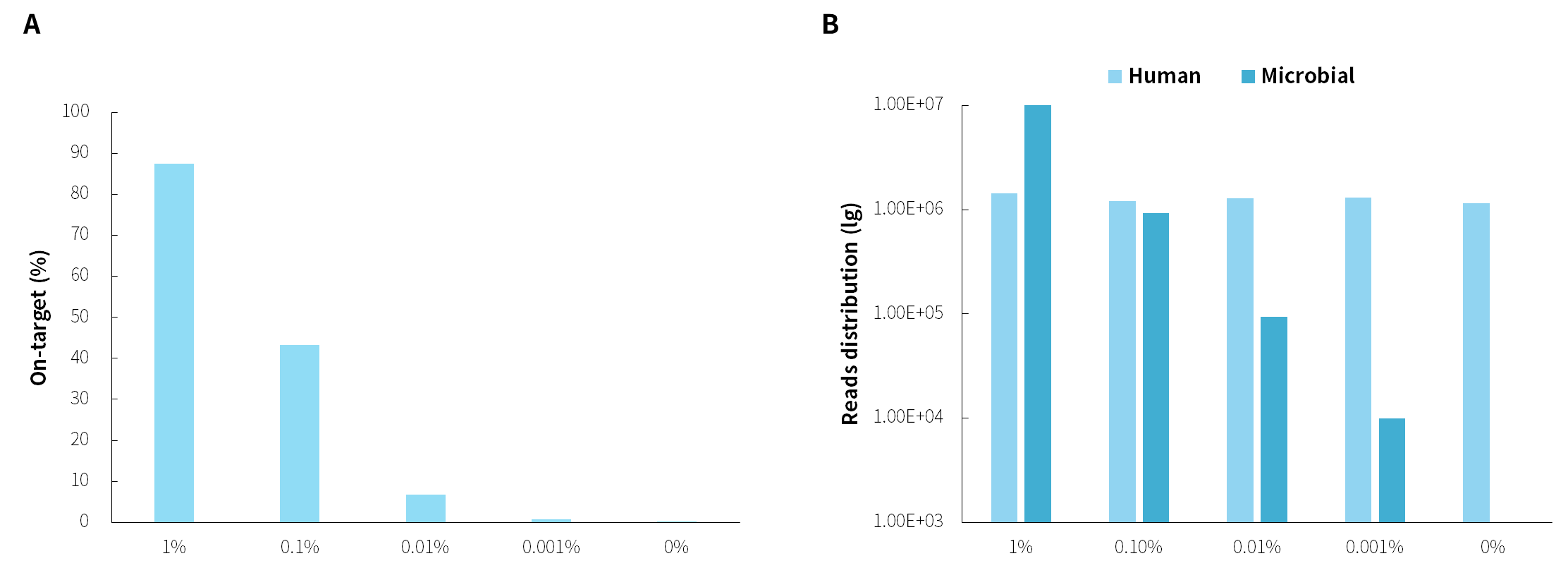 Note: Simulated microbial community samples of 0.001% - 1% MSA-1003 were created by diluting a mixture of 20 strains of genomic material (ATCC, MSA-1003) using the human genomic DNA standard (Promega, G1471) at various ratios. Library preparation was performed using 50 ng input with the NadPrep EZ DNA Library Preparation Kit v2, followed by hybrid capture (2-hour hybridization) using NEX-t Panel v1.0 with NadPrep ES Hybrid Capture Reagents. Sequencing platform: Illumina Novaseq6000, PE150. The BWA was used for alignment of raw reads to the reference genome composed of the hg19 human genome and 20 microbial genome reference sequences, and reads distribution was analyzed.
Note: Simulated microbial community samples of 0.001% - 1% MSA-1003 were created by diluting a mixture of 20 strains of genomic material (ATCC, MSA-1003) using the human genomic DNA standard (Promega, G1471) at various ratios. Library preparation was performed using 50 ng input with the NadPrep EZ DNA Library Preparation Kit v2, followed by hybrid capture (2-hour hybridization) using NEX-t Panel v1.0 with NadPrep ES Hybrid Capture Reagents. Sequencing platform: Illumina Novaseq6000, PE150. The BWA was used for alignment of raw reads to the reference genome composed of the hg19 human genome and 20 microbial genome reference sequences, and reads distribution was analyzed.
- In general, achieving complete probe coverage of pathogen microbiome genomes is impractical. On one hand, designing probes for a single bacterial species can result in hundreds of thousands of probes when considering sequence polymorphism beyond reference genomes. On the other hand, to prevent off-target effects when there is similarity between pathogenic and host sequences, certain regions must be excluded. Therefore, NEX-t Panel v1.0 emphasizes designing the fewest probes to enable the broadest range of pathogen analysis, resulting in a significantly streamlined panel size compared to tNGS schemes with millions of probes in hybrid capture.
- The number of probes can be roughly equivalent to the number of amplicons in multiplex PCR. However, due to the higher tolerance of probes and the ability to capture sequences flanking the probe, a certain number of probes can provide more diverse information compared to a similar number of multiplex amplicons.
- Compared to multiplex PCR, expanding a hybrid capture panel is simpler as it only requires adding probes. Therefore, NEX-t Panel v1.0 can be conveniently customized and upgraded through combination.

The quality of nucleic acid extraction is crucial in determining the success of high-throughput sequencing tests. Different laboratories should establish comprehensive nucleic acid extraction procedures. Firstly, the selected nucleic acid extraction reagents should be validated to ensure the efficiency and integrity of nucleic acid extraction. Quantitative measurements should be performed on each extracted nucleic acid sample to ensure that they meet the requirements for subsequent experiments. Simultaneously, standards for qualified nucleic acid samples should be established.
Nucleic Acid Quality Verification:
(1) High-quality DNA should have an A260/A280 ratio between 1.7 ~1.9, and A260/A230 > 2. DNA quality can be verified by 1% agarose gel electrophoresis (no impurities, no smearing, and no protein contamination in the background).
(2) DNA integrity should be assessed using techniques such as Agilent 2100 Bioanalyzer. If the majority of fragments are below 200 nt (except for plasma cell-free DNA, which may be 140 nt), it indicates severe DNA degradation and requires re-extraction.
(3) High-quality RNA should have an A260/A280 ratio between 1.8 ~ 2.0, with an A260/A230 ratio greater than 2.
(4) Small amounts of nucleic acid should be quantified using the Qubit fluorescence dye method.
In summary, during clinical sample collection, it is essential to ensure an adequate amount of the sample for nucleic acid extraction to obtain higher abundance nucleic acid samples. The higher the abundance of pathogenic microorganisms in the libraries prepared for tNGS, the lower the limit of detection (LOD) in tNGS detecting. This allows for efficient detection of low-load pathogenic microorganisms.
This information is primarily sourced from "Expert Consensus on the Application of High-Throughput Sequencing Technology in the Diagnosis of Mycobacterial Diseases" and "Expert Consensus on Standardizing Clinical Applications of High-Throughput Metagenomic Sequencing for Pathogen Detection."
The quality of clinical specimens directly affects the detection results. The extent to which different specimen types are influenced by colonizing microorganisms varies, leading to differences in the reliability of tNGS detection results.
The collection requirements for different types of clinical specimens
|
Type |
Volume |
DNA Sample Requirements |
RNA Sample Requirements |
||||
|
Collection Tube |
Storage |
Transportation |
Collection Tube |
Storage |
Transportation |
||
|
Blood |
≥ 10 mL |
Vacutainer with Preservation Solution |
Store at 4°C for one week |
Cold Pack Transportation |
Vacutainer with Preservation Solution |
Store at 4°C for one week |
Cold Pack Transportation |
|
Bronchoalveolar Lavage Fluid |
≥ 10 mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Sputum |
≥ 3 mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Cerebrospinal Fluid |
≥ 2 mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Pleural Fluid |
≥ 25mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Ascitic Fluid |
≥ 25 mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Bone Marrow |
≥ 0.5 mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Other Body Fluids |
≥ 10 mL |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Stool |
Size of soybean grains |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
Sterile Spiral Tube |
Long-term storage at -80°C |
Dry Ice |
|
Swab |
≥ 3 units |
Preservative Sample Tube |
Long-term storage at -80°C |
Dry Ice |
Preservative Sample Tube |
Long-term storage at -80°C |
Dry Ice |
|
Fresh Tissue |
size of rice grains |
Tissue Sampling Tube |
Long-term storage at -80°C |
Dry Ice |
Tissue Sampling Tube |
Long-term storage at -80°C |
Dry Ice |
The primary information is sourced from the Expert Consensus on the Application of High-Throughput Sequencing Technology in the Diagnosis of Mycobacterial Diseases.